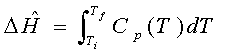So far, we've covered many Energy balance topics. Where are we?
Thus far, we have covered four energy topics. First, we covered some basic energy definitions (Ek, Ep, ΔH, and ΔU), then we learned to write energy balances on closed systems (those without streams, open systems (those with streams), and finally for mechanical systems (those where kinetic and potential energy dominate over internal energy like in for a waterfall).
For many of the rest of the problems in this class, ΔH = Q. Uh, what?
I, myself am a global learner. In 201 and in thermodynamic courses (like ChEE 316 at Arizona), this expression is first derived from an energy balance before it is used extensively in the course material after where it's connection with energy balances fades. That is, we have learned a few energy balance expressions thus far, and for the most part, we will only apply this one reduced expression for the energy balances that will come in later classes. The topics to come deal with how to find ΔH (and thus Q) with energy balances on systems of change, that is, that involve chemical reactions, as well as changes in state, temperature, pressure. So, before we begin finding the enthalpies for these changes, I would like to solidify the connection between chapter 7 and this equation, resulting in a strong connection between the energy topics of the course.
If you have just said to yourself, this is obvious, the link is clear. Chemical engineering processes usually involve flows, resulting in the use of the open energy balance. And, these processes don't usually involve valves (or other components that change the velocity -> changes in kinetic energy), or pipes involving great height changes (no potential energy), and there is often no stirrer (no shaft work). Hence, the connection between the expressions learned in chapter 7 and the ΔH = Q expression that will usually be used from here on out, is obvious. If this was you response then bravo, and you probably skip on to the interrelations of this section, if your response was "hmm", or "huh?", then take a moment, and read on to solidify the connection.
Make the connection
Keeping in mind the basic equations we learned in chapter 7, and the energy balance equations for closed, open, and mechanical systems, let's consider a simple chemical process where methanol and water are separated in a condenser. What energy equations would you apply to the following system? How?
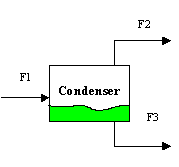
So, consider your energy balance tool box: Which tool do you use, and how? Well, in the tool box we have a closed energy balance, an open one, and a mechanical system energy balance equation. There are streams in this problem (flowrates), thus the system is not closed. There are no valves or huge height changes so the system is not a strictly mechanical system using only a mechanical energy balance. Thus, it is clearly an open system where only the open energy balance equation applies. Finally, the equation is:
ΔH+ΔEk+ΔEp=Q+Ws
ΔH+ΔEk+ΔEp=Q+Ws
ΔH=Q