Solution 3.2
|
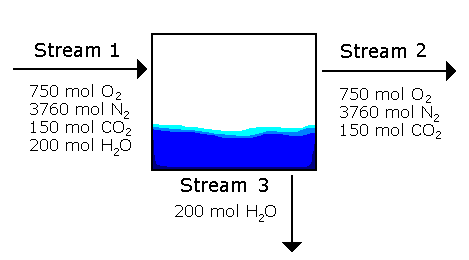
Example 3.3 Problem Statement:
The condenser has one inlet and two outlets, but only one inlet and one outlet contain mixtures of compounds.
The bottom outlet contains only water, so yH2 = 1.0, the mole fraction of water is 1.0.
For the other two streams, the mole fraction of each compound can be calculated by knowing the total amount of moles in the system. Remember, yA is the mole fraction of A in the mixture, which is defined as:
For the two streams the total moles must be calculated first. For the inlet stream:
nTOTAL = 4860 moles total The mole fractions of each compound in the inlet stream can now be calculated:
yO2 = 0.1543 yN2 = 3760 moles N2 / 4860 moles Total yN2 = 0.7737 yCO2 = 150 moles CO2 / 4860 moles Total yCO2 = 0.0309 yH2O = 200 moles H2O / 4860 moles Total yH2O = 0.0412
yTOTAL = 1.0001 For the outlet stream, the total number of moles can be calculated:
nTOTAL = 4660 moles total
yO2 = 0.1609 yN2 = 3760 moles N2 / 4660 moles Total yN2 = 0.8069 yCO2 = 150 moles CO2 / 4660 moles Total yCO2 = 0.0322
yTOTAL = 1.0000 Both sets of mole fractions add up to 1.0, so our answers for each stream are reasonable. |

The University of Arizona. All copyrights © reserved.